The U.S. health innovation ecosystem stands as a beacon of progress, influencing global standards in biomedical innovation. Emerging from pivotal moments in history, particularly during World War II, this ecosystem catalyzed breakthroughs like the mass production of penicillin—an early triumph of public-private research partnership. Federal funding in research has since played a crucial role in propelling advancements in medicine and technology, fostering an environment rich in innovation. As policymakers navigate contemporary challenges, understanding the historical impact of the war on science highlights the importance of sustained support for federal initiatives. With a thriving network of universities, research institutions, and private companies, the U.S. continues to lead the way in developing transformative healthcare solutions.
The American healthcare innovation landscape is a complex and dynamic arena that has garnered attention due to its remarkable achievements in medical technology and research. Rooted in the collaborative efforts of academia, private sector companies, and government funding, this system has evolved significantly since its inception. The post-World War II era marked a significant turning point for biomedical research, as it introduced federal programs that facilitated the growth of novel therapeutics and enhanced public health initiatives. Partnerships between various stakeholders have enabled the rapid translation of scientific discoveries into practical applications, essential for addressing current health challenges. As we reflect on this rich history of U.S. advancements, it becomes clear how integral these collaborations are for future breakthroughs in health innovation.
The Foundation of the U.S. Health Innovation Ecosystem
The U.S. health innovation ecosystem is rooted in a complex history that intertwines with global events, notably World War II. At the time, the demand for innovative medical solutions surged as infectious diseases were responsible for more soldier fatalities than battlefield injuries. This dire need led to an unprecedented collaboration between government and academia, establishing a model that has remained relevant. The mass production of penicillin stands as a key milestone, illustrating how federal funding and academic research can effectively address immediate public health challenges while also laying the groundwork for future biomedical advances.
As the war progressed, the Office of Scientific Research and Development (OSRD) emerged as a crucial player in coordinating wartime research efforts. New strategies, such as forming public-private research partnerships, not only facilitated the rapid development of life-saving technologies but also set a precedent for ongoing collaboration. The lessons learned and the relationships forged during this period have fostered a unique innovation ecosystem that has propelled advancements in biomedicine, ensuring a continuous pipeline of research and development that benefits both military and civilian health.
Public-Private Research Partnerships in Biomedical Innovation
The partnership between government entities and private research institutions has been instrumental in nurturing the U.S. biomedical innovation landscape. Initially spurred by the urgent demands of the war, this collaboration evolved into a robust framework that has driven technological advancements and discoveries in medicine. The public-private partnership model enables pooling of resources, expertise, and funding, resulting in synergy that accelerates innovation. By leveraging federal funding initiatives, such as those from the National Institutes of Health (NIH), academic institutions have been able to explore novel research avenues, thus expanding the frontiers of biomedical science.
However, the sustainability of these partnerships faces scrutiny, particularly in light of evolving federal funding landscapes. Recent proposals to cap reimbursements for research costs could threaten the delicate balance that underpins these collaborations. Researchers argue that maintaining a supportive funding structure is necessary to continue achieving breakthroughs in biomedicine. As history has shown, when the government invests in scientific research, it not only benefits national defense and health but also stimulates economic growth, establishing a stable environment that fosters innovation across the biotech industry.
The Impact of World War II on U.S. Innovation
World War II catalyzed significant advancements in U.S. innovation, particularly in biomedical research. The urgency to solve pressing health issues, like infectious diseases plaguing soldiers, led to foundational changes in research practices and organizational structures. Federal agencies redefined the role of government in technology development by facilitating collaborations that brought together academia, industry, and military needs, establishing a precedent for sustained innovation. This period marked a transformative era, paving the way for what we recognize today as a thriving U.S. health innovation ecosystem.
The legacy of these wartime innovations continues to influence contemporary practices in biomedical research. The systems established during this time encouraged rigorous scientific inquiry, bolstered by substantial federal funding and support. For instance, the introduction of peer review processes and funding policies during the war years fostered a culture of meticulous research practices that remain critical to the field today. As we look back, it is evident that the innovations born out of necessity during World War II have had lasting impacts on public health and continue to shape the trajectory of U.S. science and technology.
Evolution of Federal Funding in Research
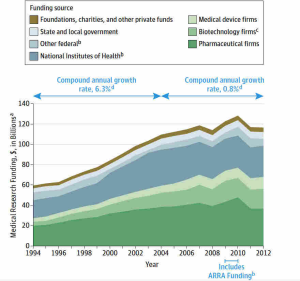
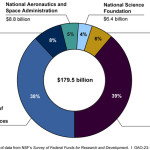
Federal funding has played a pivotal role in the evolution and success of the U.S. health innovation ecosystem. Historically, federal investment has enabled universities and research institutions to thrive, driving forward groundbreaking discoveries in biomedical science. The establishment of organizations like NIH has empowered researchers to convert theoretical knowledge into practical medical advancements. This funding model is not only essential for supporting current research but also for training the next generation of scientists who will inherit and expand upon this legacy.
The debate surrounding federal funding, particularly concerning reimbursement for indirect costs, reflects ongoing challenges within the innovation system. While there is a strong rationale for maintaining robust investment in biomedical research, recent discussions highlight the need for balance between accountability and ensuring that research institutions can effectively operate without financial hindrance. Navigating these complexities is vital for preserving the innovative spirit that has characterized U.S. biomedical research and ensuring continued advancements that benefit society as a whole.
Historical Context of U.S. Biomedical Innovation
Understanding the historical context of U.S. biomedical innovation enhances our appreciation of the current health innovation ecosystem. The post-World War II era witnessed an explosion of research fueled by federal funding and the collaborative efforts of academic institutions and industry. Innovations such as the development of antibiotics and vaccines became milestones of this era, saving countless lives and transforming public health outcomes. The transition from wartime urgency to peacetime promotion of science laid the groundwork for a culture of persistent innovation that is still relevant today.
Furthermore, the history of U.S. innovation also underscores the importance of adaptability and resilience in research practices. As challenges in public health evolve, so too must the frameworks that support innovation. The partnerships formed during pivotal moments in history—specifically during crises like World War II—have established a robust foundation that has allowed for rapid responses to emerging health threats. This historical perspective encourages current policymakers and researchers to build on established successes and adapt to the changing landscape of biomedical research.
The Role of Government in Biomedical Research
The government has played an integral role in shaping the U.S. biomedical research landscape. Historically, federal support has been crucial in funding research initiatives that drive innovation in health and medicine. Government-led efforts during World War II exemplified a proactive approach to addressing public health needs by establishing agencies dedicated to scientific research and innovation, catalyzing advancements that can be traced to today’s cutting-edge biomedical research. This government involvement is not merely transactional; it is foundational, creating an ecosystem where innovation can flourish.
In recent years, as discussions about federal funding shift in response to changing priorities, the importance of sustained government support cannot be overstated. The intricate relationship between government and research institutions is essential for navigating complex challenges, such as emerging diseases and healthcare delivery inefficiencies. Future investments must honor the historical commitment to driving biomedical innovation, ensuring that researchers are empowered to harness new possibilities and tackle pressing health issues head-on.
Strengthening U.S. Health Innovation for the Future
As we look to the future of the U.S. health innovation ecosystem, it is essential to build upon the solid foundations laid in the past. Policymakers, researchers, and industry leaders must collaborate to create a framework that encourages innovation while addressing contemporary challenges. This requires an understanding of the intricate dynamics of public-private partnerships and a commitment to continued investment in biomedical research—a critical pillar that has traditionally driven health advancements.
Strategies to ensure ongoing support for biomedical innovation could include advocating for increased federal funding, streamlining grant processes, and fostering educational initiatives that prepare the next generation of scientists. Emphasizing the importance of interconnectedness within the ecosystem will help maintain the motivational momentum fueled by historical successes—enabling the U.S. to remain at the forefront of global health innovation and ensuring that future generations benefit from enhanced medical technologies and improved health outcomes.
Impact of the COVID-19 Pandemic on Biomedical Innovation
The COVID-19 pandemic underscored the critical importance of a robust U.S. health innovation ecosystem, revealing both strengths and weaknesses in the system. It prompted an accelerated pace of research, resulting in unprecedented vaccine development and a renewed focus on public health preparedness. The collaborative architecture established during historical moments—such as World War II—provided a framework for rapid mobilization of resources, showcasing the responsiveness of U.S. biomedical innovation in the face of global crises.
As researchers and industry leaders reflect on the lessons learned from the pandemic, there is a growing recognition of the need for sustained investment and collaboration. Addressing future health threats will require flexibility and continued engagement within the public-private research partnerships that have defined U.S. innovation. By championing a culture of resilience and adaptability, the biomedical community can ensure that it is well-equipped to tackle not only the current challenges but also those on the horizon.
Future Challenges for Biomedical Research Funding
Despite the historical successes of the U.S. health innovation ecosystem, future challenges loom large in the arena of biomedical research funding. The increasing complexity of health issues and the pressure on federal budgets pose significant obstacles, particularly as policymakers contemplate caps on reimbursements and allocate resources unevenly. Maintaining the balance between supporting innovative research and managing budgetary constraints is essential for the continued vibrancy of the biomedical sector.
Navigating these challenges requires a concerted effort from various stakeholders, including government agencies, academic institutions, and industry players. By fostering transparent dialogues and reimagining funding frameworks, the ecosystem can ensure that adequate support remains available for critical research initiatives. Protecting the public-private research partnership model will be vital for preserving the legacy of U.S. biomedical innovation and enabling a future characterized by significant health advancements.
Frequently Asked Questions
What is the U.S. health innovation ecosystem and how did it originate?
The U.S. health innovation ecosystem is a collaborative network involving universities, the life sciences industry, and federal research funders, primarily represented by the National Institutes of Health (NIH). Its origins can be traced back to World War II, when federal funding and public-private research partnerships were established to drive technological advancements in response to wartime needs, such as the mass production of penicillin. This partnership laid the groundwork for the innovative biomedical research landscape we see today.
How did World War II impact the U.S. health innovation ecosystem?
World War II served as a catalyst for the U.S. health innovation ecosystem by highlighting the urgent need for medical advancements to protect soldiers. The government initiated the Office of Scientific Research and Development (OSRD), which fostered public-private research partnerships and increased federal funding for biomedical research. This collaboration not only addressed immediate health concerns but also spurred groundbreaking developments in biomedicine that continued to influence the ecosystem in the decades that followed.
What role does federal funding play in the U.S. health innovation ecosystem?
Federal funding is critical to the U.S. health innovation ecosystem as it supports academic research, combines public-private research partnerships, and facilitates technological advancements in medicine. The NIH, through various grants and funding initiatives, provides essential financial backing that encourages innovation and development in the biomedical sector, ensuring a continuous pipeline of new discoveries and treatments.
What are some key advancements from the U.S. health innovation ecosystem since World War II?
Since World War II, the U.S. health innovation ecosystem has produced numerous key advancements, including the development of antibiotics like penicillin, vaccines, advanced surgical techniques, and personalized medicine. The collaboration between the federal government and academic institutions has led to the creation of new therapies and technologies, significantly improving patient outcomes and enhancing the overall quality of healthcare.
What challenges does the U.S. health innovation ecosystem face today?
Today, the U.S. health innovation ecosystem faces challenges such as potential reductions in federal funding due to changing political climates, pressures to cap reimbursements for indirect research costs, and ensuring systemic efficiency without undermining the established partnerships. As the ecosystem evolves, maintaining a balance between public funding, private investment, and innovation is vital to sustaining its global leadership in biomedicine.
How do public-private research partnerships enhance the U.S. health innovation ecosystem?
Public-private research partnerships enhance the U.S. health innovation ecosystem by fostering collaboration between government agencies, academic institutions, and private industry. This synergy leads to shared resources, expertise, and risk, accelerating the development of new medical technologies and treatments. By pooling knowledge and funding, these partnerships have been instrumental in driving significant advancements in biomedical research and innovation.
What is the historical significance of U.S. biomedical innovation post-World War II?
The historical significance of U.S. biomedical innovation post-World War II lies in its profound impact on global health standards, military medicine, and the pharmaceutical industry. The collaboration fostered during the war laid the foundation for an enduring research culture, facilitating breakthroughs in drug development, public health strategies, and innovative medical technologies that have shaped modern healthcare systems worldwide.
| Key Points |
|---|
| The U.S. health innovation ecosystem is recognized globally for its effectiveness. |
| Originated during WWII with government-backed research leading to mass production of penicillin. |
| Public-private partnerships have been critical to advancements in biomedical science. |
| Historically, infectious diseases posed significant challenges, prompting urgent innovation. |
| Foundation laid by wartime initiatives continues to support postwar biomedical advancements. |
| Federal funding remains a vital component, enabling research and development across multiple sectors. |
| The unique collaborative model among academia, industry, and government has fostered persistent success. |
| Maintaining this synergy is crucial for future growth and to guard against funding cuts. |
Summary
The U.S. health innovation ecosystem stands out as a model of success worldwide, characterized by its deep-rooted public-private partnerships formed during pivotal moments such as World War II. These collaborations have not only facilitated significant medical advancements but have also created a sustainable system that continues to thrive today. Given its historical significance and ongoing impact, preserving this ecosystem is essential for safeguarding future breakthroughs that benefit both the nation and the global community.